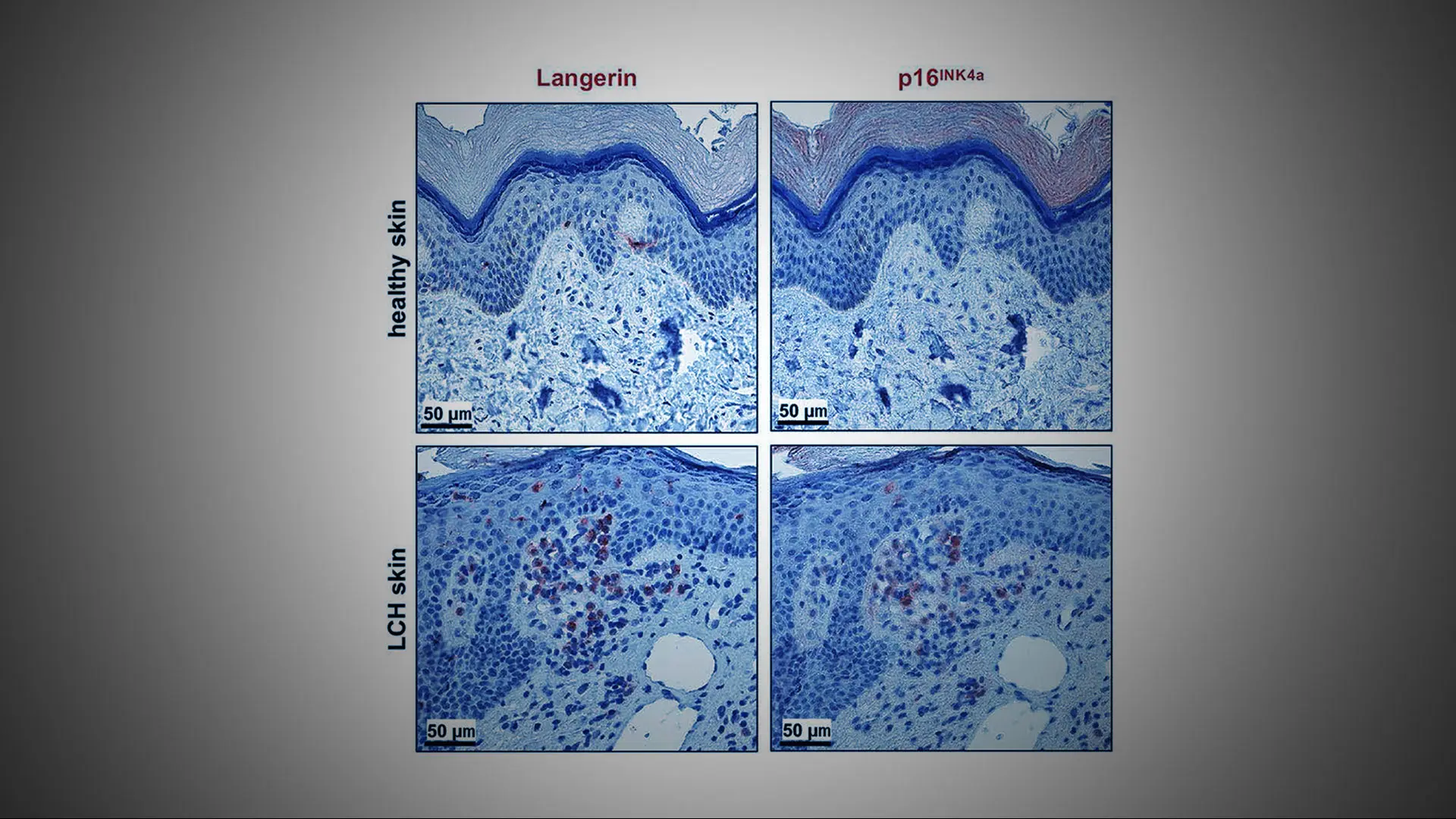
A 17-year quest to understand Langerhans-cell histiocytosis (LCH)—an inflammatory blood disease that mostly effects children and can result in dementia and death—has led researchers at the Icahn School of Medicine at Mount Sinai and their colleagues in Texas to a transformational discovery and a potential new treatment. Their findings were published in May in Nature Medicine.
The team, led by Miriam Merad, MD, PhD, Director of the Precision Immunology Institute at Icahn Mount Sinai, and Carl E. Allen, MD, PhD, Professor of Pediatrics, Hematology-Oncology, at Baylor College of Medicine, found that individuals with LCH have a mutation that puts a
subset of white blood cells into a state called senescence. In this state, the cells stop multiplying as they normally would, express a pro-survival gene, and begin producing inflammatory molecules.
Lesions, a hallmark of the disease, form wherever the inflammation occurs, on the skin and in various organs including the brain and central nervous system, manifesting differently in each patient and causing a range of severity. Roughly one in 100,000 children in the United States
develops LCH each year, putting it on par with the incidence of pediatric Hodgkin lymphoma, the third most common cancer in children. LCH appears to be most prevalent in the Middle East and countries such as China.
Understanding the role senescence plays in LCH has enabled the researchers to find a potential treatment that “attacks the cause of the problem,” says Dr. Merad. “We have a therapeutic molecule that can get at this pathway and block the ability of these senescent cells to survive.” When the researchers tested the molecule in animal models and on human cells they found the diseased cells were “super-addicted to this survival signal. So now
we have a way of blocking the survival signal.”
LCH is not inherited and appears to have only one mutation.
Plans are underway, she adds, to launch an international, multi-site clinical trial in September, in conjunction with a pharmaceutical sponsor and the National Institutes of Health. Details will be released when the contracts are formalized.
If successful, Dr. Merad says the therapeutic molecule would replace the use of chemotherapy, the current standard of care, which fails to work in the majority of cases. Chemotherapy helps to eliminate some of the inflammation, she says, but it also kills healthy cells and is unable to reach the diseased cells.
Even with the latest breakthrough, LCH remains a baffling disease. It is not inherited, and there appears to be only one mutation. “It’s very unique,” says Dr. Merad. “We don’t know the cause or what makes some kids more at risk.” Upcoming clinical trials should help shed more
light on the disease mechanisms.
Drs. Merad and Allen have been collaborating on LCH research since 2004, when they met at a conference in Greece, which was sponsored by the father of a child with a severe case of LCH, who was frustrated by the lack of scientific knowledge. Five years later, Drs. Merad
and Allen were the first to describe LCH as a disease of the hematic system—blood vessels that carry blood throughout the body—which originates in the bone marrow. Prior to that, LCH was categorized as a type of skin cancer.
The next piece of the puzzle is preventing neurodegeneration.
“This disease should be classified as an inflammatory hematological disease,” says Dr. Merad. “There’s no proliferation; the cell never metastasizes. What we’ve discovered in this latest paper is that the mutation puts the cells into a state that is not cancerous, but one in which
the cells are fighting not to expand. They start producing a lot of inflammatory molecules because they sense there is danger. This inflammation is the cause of most of the symptoms.”
She says the next piece of the LCH puzzle will be figuring out how to prevent the disease from causing neurodegenerative damage.
The 17-year journey to reach this latest discovery has been a “story of friendship, commitment, and perseverance,” says Dr. Merad. It was her mentor, the late Nobel laureate Ralph M. Steinman, MD, who first asked her to join him in studying LCH and brought her to the 2004 conference where she met Dr. Allen. She became committed to understanding the baffling disease after being greeted at the door by the organizer’s teenage son, who had LCH, and by his father, who had enlisted some of the world’s leading scientists to find answers.
“What’s beautiful is that this group we formed in 2004 (with Dr. Allen’s lab) is as strong today as it was then,” says Dr. Merad. “We never stopped working on these questions. Science takes perseverance. We start with the question, and we dig and dig until we have something solid and then we put it out there. No matter what it takes we continue to work on it.”
Featured

Miriam Merad, MD, PhD
Director of the Precision Immunology Institute, and the Mount Sinai Human Immune Monitoring Center