Twenty years ago, Judy H. Cho, MD, helped identify the first genetic abnormality linked to Crohn’s disease—loss-of-function variants in the microbial-sensing domain of the NOD2 gene. Since then, Dr. Cho, Dean of Translational Genetics, and Director of The Charles Bronfman Institute for Personalized Medicine at the Icahn School of Medicine at Mount
Sinai, has steadily advanced research into this inflammatory intestinal disease, work that led to her election in 2020 to the National Academy of Medicine.
Recently, Dr. Cho and a team of investigators at Icahn Mount Sinai discovered a cellular mechanism that prevents a subset of Crohn’s patients from responding to a widely used treatment for moderate and severe cases: anti-tumor necrosis factor (TNF) inhibitors. In a study that was published in the March 31, 2021 issue of Nature, the researchers reported
that blocking the common cytokine receptor subunit gp130 may benefit these unresponsive
patients. TNF inhibition medication blocks white blood cells that produce the protein TNF, which causes the inflammation.
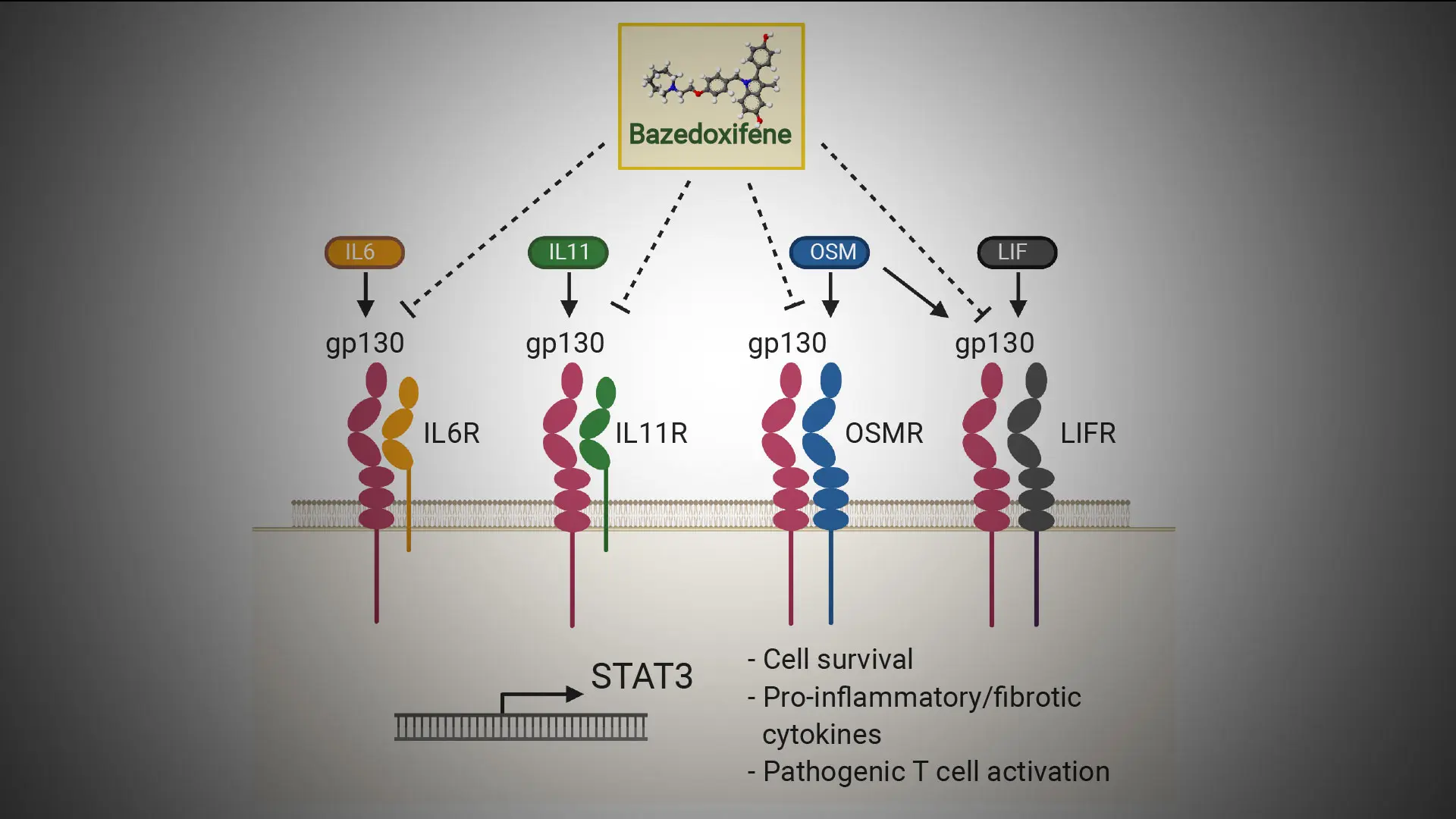
gp130
Blocking this cytokine receptor subunit may help unresponsive patients
The Mount Sinai researchers analyzed inflamed and normal tissues of the small intestine in humans and zebrafish models of intestinal injury. They showed that patients carrying Crohn’s-associated mutations in the NOD2 gene have increased activated fibroblast and macrophage gene expression, and, in particular, elevated gp130-related gene expression.
“Our work defines a completely new mechanism whereby NOD2 mutations confer risk, namely through altered differentiation of newly recruited blood monocytes over time,” says Dr. Cho. “It sharpens current research efforts involved in serial tissue and blood analyses to define how non-response or loss of response to anti-TNF therapies may be improved.”
Shikha Nayar, PhD, the study’s first author and a Postdoctoral Fellow in Dr. Cho’s lab at Icahn Mount Sinai, says the findings could provide a more custom-made approach to future patient care. “We’ve developed novel in vivo and in vitro models to define mechanisms and timing of disease progression,” she says. “These studies may tailor treatments more effectively
for Crohn’s patients carrying NOD2 mutations and elevated signatures we have described."
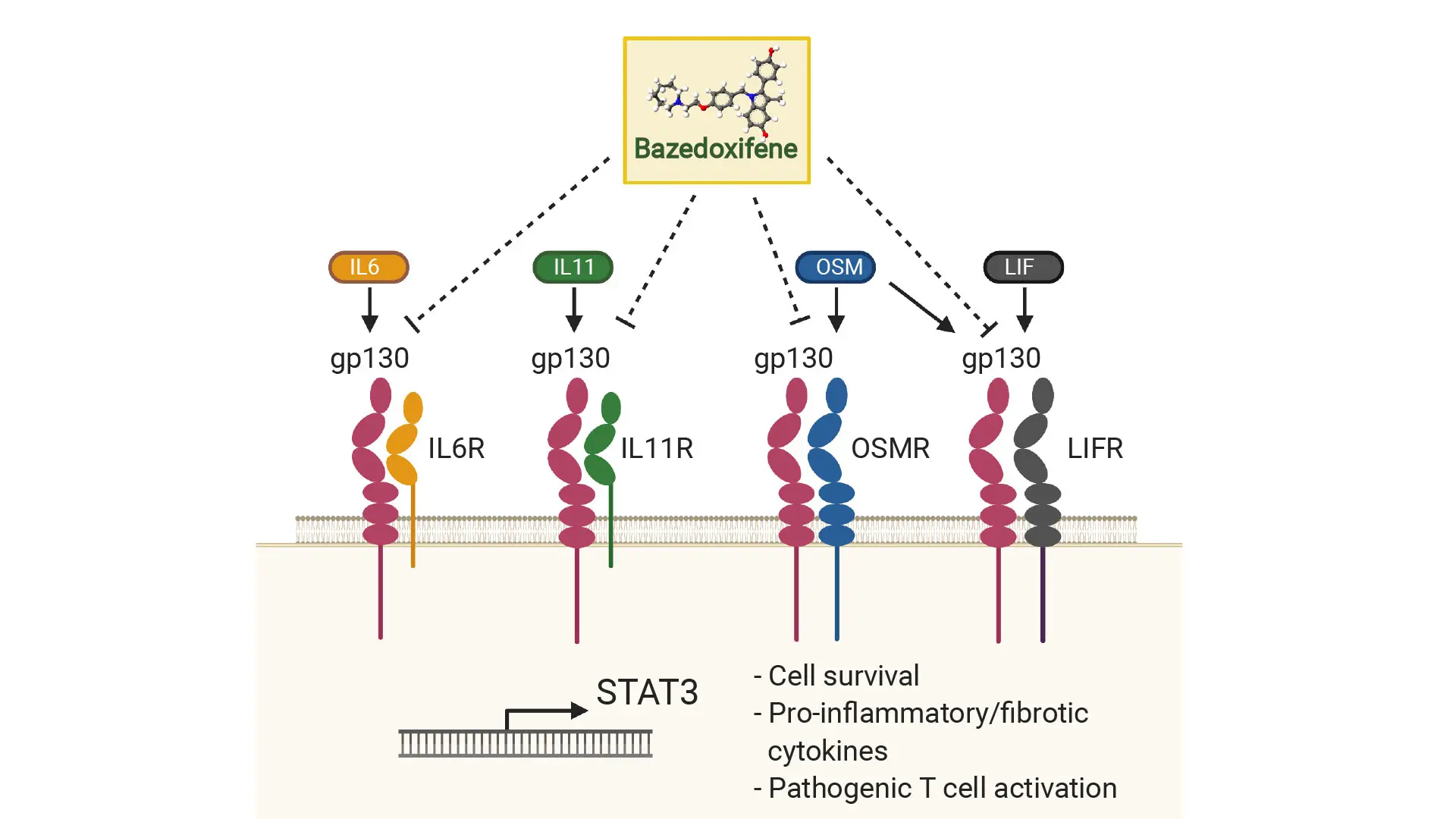
As shown here, gp130 is a common receptor for its family of receptor genes (IL6R, IL11R, LIFR, OSMR). To resolve cellular activation in Crohn's disease, bazedoxifene, a U.S. Food and Drug Administration-approved drug, could effectively inhibit gp130 and its partner receptors and potentially serve as a complementary treatment for patients who do not respond to anti-TNF therapy.
Featured

Judy H. Cho, MD,
Dean of Translational Genetics, and Director, The Charles Bronfman Institute for Personalized Medicine

Shikha Nayar, PhD,
Postdoctoral Fellow